Innovation Passport Granted for CTx001 Gene Therapy in Geographic Atrophy
- MHRA granted Complement Therapeutics an Innovation Passport under the new accelerated Innovative Licensing and Access Pathway (ILAP)
- ILAP is designed to optimise the development and expedite the evaluation of innovative medicines for patients with high unmet need
- CTx001 is being evaluated as a potential AAV gene therapy for the treatment of visual impairment due to geographic atrophy
Complement Therapeutics Ltd (CTx), a biotechnology company developing novel therapeutics for complement-related diseases, today announced its lead asset CTx001, an AAV-based gene therapy for the treatment of Geographic Atrophy (GA), has been awarded an Innovation Passport by the UK’s Medicines and Healthcare Products Regulatory Agency (MHRA).

CTx001 Recognised for Innovation and Potential in Treating GA
CTx001 was awarded the Innovation Passport based on three criteria: that the condition addressed is seriously debilitating with a significant patient need; an advanced therapy medicinal product classified as innovative; and that the medicine has the potential to offer transformative benefits to patients.
“Receiving the Innovation Passport Designation is a key regulatory accomplishment and has the potential to help GA patients in the UK gain faster access to our novel gene therapy. It provides recognition from the MHRA of the unique mechanism of action of CTx001, with the potential to change the treatment landscape for GA patients. We look forward to working with the MHRA and other agencies to advance the CTx001 development programme.” – Dr Rafiq Hasan, CEO of Complement Therapeutics
Benefits of ILAP for Accelerated Gene Therapy Development
The Innovation Passport is the first step in the Innovative Licensing and Access Pathway (ILAP) by providing opportunities for enhanced regulatory support from the MHRA and its partner agencies, including the National Institute for Health and Care Excellence (NICE), the Scottish Medicines Consortium (SMC) and National Health Service (NHS) England. CTx will work with these agencies on a Target Development Profile (TDP) for CTx001, that will set out a roadmap for regulatory and development milestones to enable early patient access in the UK. Other benefits of ILAP, include the potential for a 150-day accelerated Marketing Authorisation Application (MAA) assessment, a rolling review and a continuous risk-benefit assessment.
About the Innovative Licensing and Access Pathway (ILAP)

The MHRA launched ILAP at the start of 2021 in order to accelerate the development and access to promising medicines in the early stages of development. It provides a single integrated platform for collaboration between the developer of the investigational medicine, the MHRA and its partners. The pathway, part of the UK’s plan to attract life sciences development in the post-Brexit era, features enhanced input and interactions with MHRA and other stakeholders.
More background on the ILAP can be found at: https://www.gov.uk/guidance/innovative-licensing-and-access-pathway
About Complement Therapeutics Ltd

Complement Therapeutics (CTx) is an early-stage biotechnology company focused on the research and development of novel therapeutics for complement-mediated diseases. The company is a spinout from the University of Manchester and is based on the pioneering research of its founders into novel targets within the complement cascade.
Our lead investigational product (CTx001) is being evaluated as a potential gene therapy for dry age-related macular degeneration/geographic atrophy, a leading cause of blindness. Additional programmes will evaluate potential therapeutic opportunities in other complement-mediated conditions.
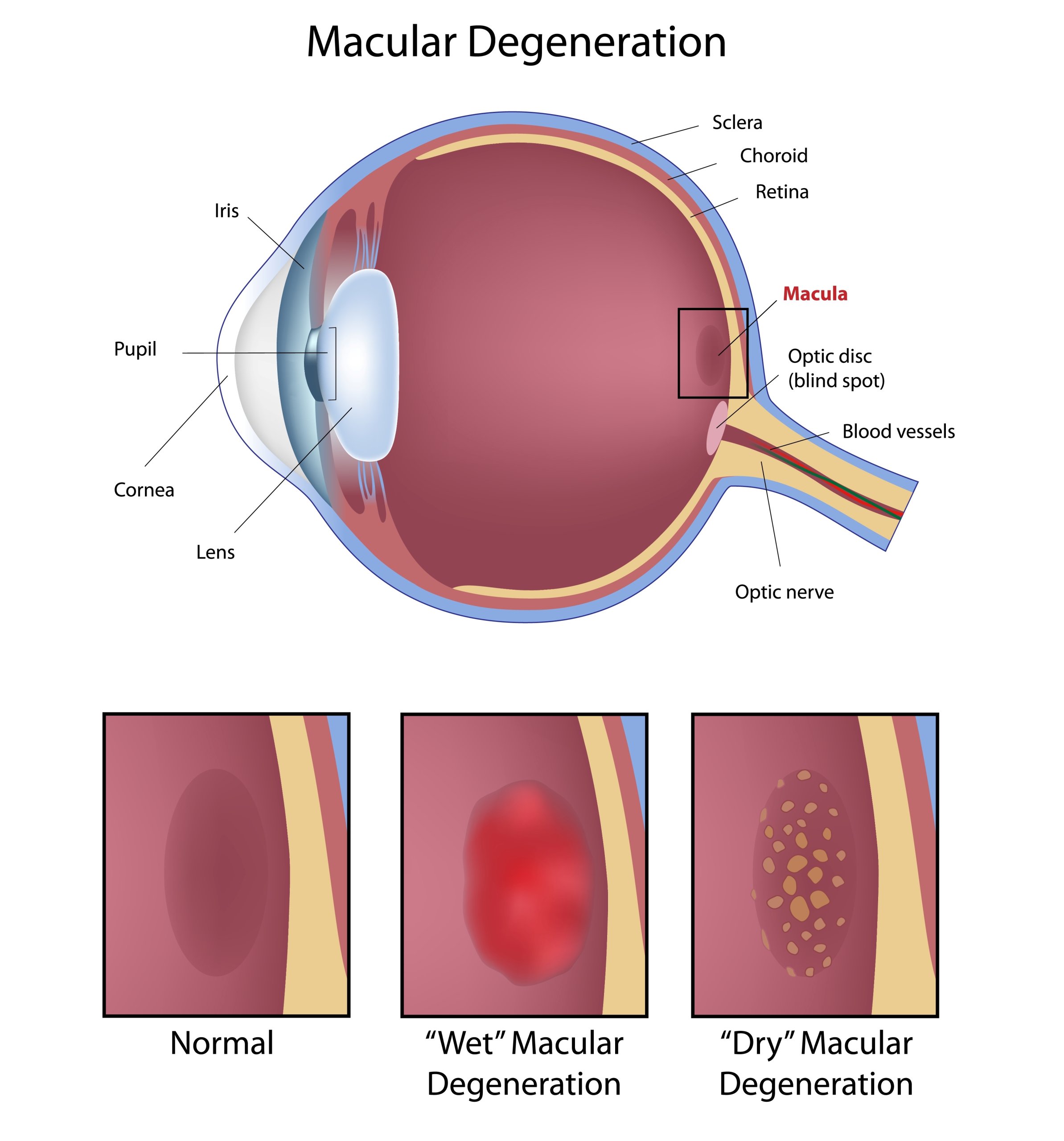
Eye condition: macular degeneration
The Company is also developing a unique quantification methodology, the Complement Precision Medicine (CPM) platform, to enable quantification of over 30 complement cascade proteins enabling more precise diagnosis and monitoring of disease.
To find out more about Complement Therapeutics, contact clare.arkwright@uominnovationfactory.com